Sysmex and ESR collaborate on national COVID-19 results CDR
Within hours of New Zealand lifting to alert level 4, a collaborative project was initiated between the Institute of Environmental Science and Research (ESR) and Sysmex New Zealand to establish a national clinical data repository (CDR) for all COVID-19 test reports from testing lab facilities across the country.
ESR provide an extensive range of scientific services and research capability and are the public health laboratory for all notifiable diseases, including COVID-19. The repository complements EpiSurv, the system ESR operates for the Ministry of Health on all notifiable disease information from the Public Health Services (PHS) in New Zealand.
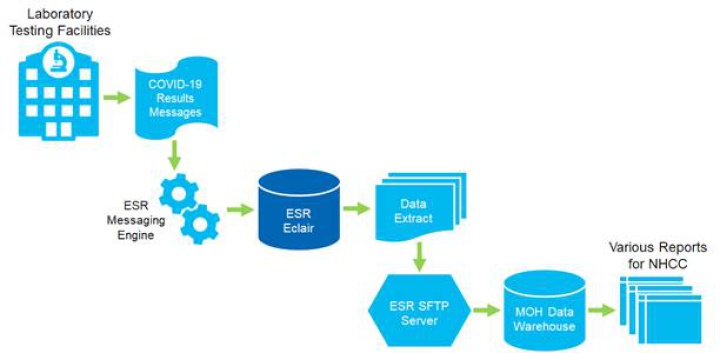
To effectively manage New Zealand’s response to the COVID-19 pandemic, there emerged an urgent need for national level COVID-19 lab testing data to be rapidly collected, collated and analysed by the team at ESR. Laboratory test facilities across NZ were tasked with collating and forwarding this data to ESR for loading into a data warehouse. This information was then used in intelligence reporting for the National Health Coordination Centre (NHCC) and the Ministry to use in the daily COVID-19 update to the nation.
“As with much of New Zealand, COVID-19 has produced unique challenges for the systems our country has,” explains ESR Data and Informatics Manager Andrew Crooke. “The speed at which we need data at the moment is unprecedented and we wanted to find a way to streamline that data for both the laboratories in New Zealand and the Ministry of Health. So, we teamed up with Sysmex New Zealand and the laboratories across New Zealand to find solutions to get data faster and more easily to the Ministry of Health.”
Sysmex New Zealand’s Eclair CDR is a proven and trusted solution for the standardised storage, management and display of diagnostic and clinical data by District Health Boards and private pathology providers across New Zealand and overseas.
Laboratory testing facilities maintain their standard notification process for positive samples to EpiSurv but have updated their reporting process to also provide test results via an HL7 message for every sample, whether it be positive or negative, to ESR. This real-time lab testing data is received and stored in ESR’s Eclair CDR and can be easily extracted to provide a full and complete national view of COVID-19 testing. This, in turn, means quicker and easier access to testing volumes and the percentage of positive results versus number of tests undertaken on a daily or ad-hoc basis.
The importance of the situation has led to some extraordinary moments during this project, including the speed at which the Eclair database was installed. While the usual security protocols when working with ESR and the Ministry were strictly followed, they were undertaken with a degree of urgency, allowing the Sysmex New Zealand Eclair and ESR teams to rapidly setup hardware configuration and deploy the Eclair database.
In addition to providing a national database for all COVID-19 test reports, ESR’s Eclair CDR delivers an easily accessible and more robust lab testing results management solution for ESR and takes the pressure of New Zealand laboratories to collate this information manually every day.


 University of Auckland: Research To Address Equity In STEM For Māori, Pacific And Female Students
University of Auckland: Research To Address Equity In STEM For Māori, Pacific And Female Students Stats NZ: Economic Impacts On New Zealand From Conflict In The Middle East – Report
Stats NZ: Economic Impacts On New Zealand From Conflict In The Middle East – Report Advertising Standards Authority: ASA Annual Report 2025 - Platform-Neutral Regulation Keeps Pace With Digital Advertising
Advertising Standards Authority: ASA Annual Report 2025 - Platform-Neutral Regulation Keeps Pace With Digital Advertising Science Media Centre: Lead Pipes Banned For New Plumbing – Expert Reaction
Science Media Centre: Lead Pipes Banned For New Plumbing – Expert Reaction New Zealand Young Physicists Trust: Auckland To Host The ‘World Cup Of Physics’ In 2027; Search Begins For Student-Designed Tournament Logo
New Zealand Young Physicists Trust: Auckland To Host The ‘World Cup Of Physics’ In 2027; Search Begins For Student-Designed Tournament Logo Oxfam Aotearoa: Top CEO Pay Increased 20 Times Faster Than Workers’ Pay In 2025
Oxfam Aotearoa: Top CEO Pay Increased 20 Times Faster Than Workers’ Pay In 2025


