MARS Bioimaging Mobile Photon-Counting CT Scanner Selected For Contrast Agent And AI Research
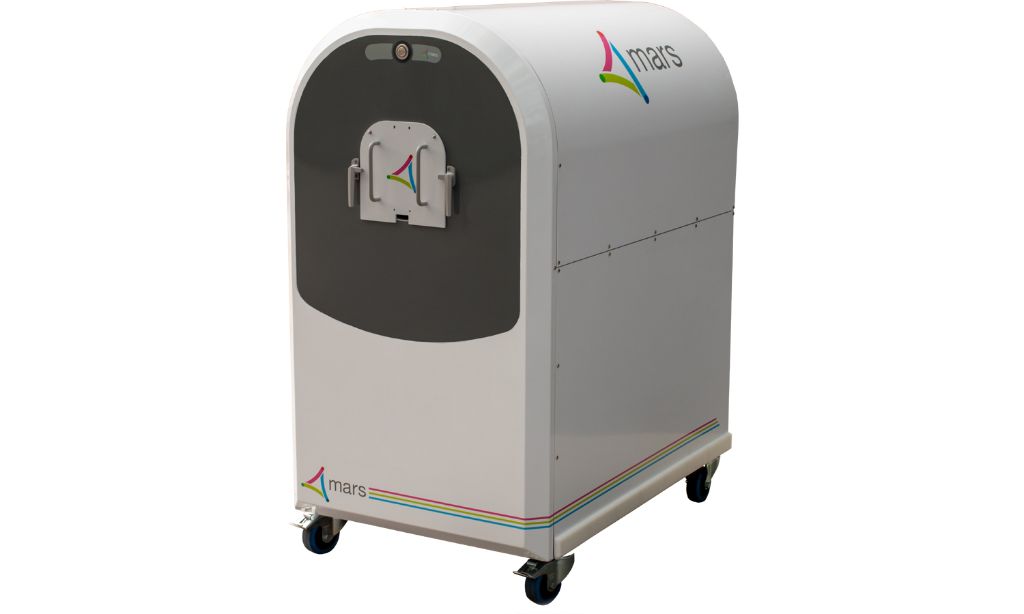
CHRISTCHURCH, NZ, May 13, 2026 – MARS Bioimaging, a New Zealand–headquartered medical device company, today announced that it has sold a mobile photon-counting CT scanner to Bracco Imaging S.p.A., a global leader in diagnostic imaging, to support its contrast agent and artificial intelligence research.
The MARS Bioimaging Scanner System will be installed at Bracco’s Research Center (CRB) in Colleretto Giacosa, Italy, a facility focused on the development and innovation of contrast agents for X-ray/CT and MR imaging.
Bracco Imaging lead investigator, Alberto Fringuello Mingo, said: “The acquisition of the MARS photon-counting CT scanner is supported by ‘Fondo Italiano per le Scienze Applicate’ funded research project (grant: FISA-2023-00298), led by Principal Investigator Prof. Angelo Bifone of the University of Turin. This project will investigate how AI and advanced imaging can be combined to enhance diagnostic outcomes, also leveraging the unique capabilities of photon-counting CT—such as spectral imaging and material decomposition—to achieve improved characterization of contrast.”
Dr. Ojas Mahapatra, Group CEO, MARS Bioimaging, said: “We’re excited that a world leader in diagnostic imaging and a trusted partner for radiologists, has selected our technology to support its research. This sale to a leading industry innovator represents an important commercial milestone and signals growing momentum for MARS.”
The MARS Bioimaging Extremity Scanner System received U.S. Food and Drug Administration (FDA) 510(k) clearance in March 2026, providing important validation to support regulatory pathways and market adoption globally.
Mark Figgitt, Group COO, MARS Bioimaging, added: “MARS scanners deliver high-resolution 3D X-ray imaging that visualises soft tissue, bone, blood vessels and metallic implants with clear material differentiation, enabling new insights into how contrast agents can enhance diagnostic performance. Importantly, researchers can also readily access underlying data in an efficient workflow designed to add value to clinical research projects.”
The MARS scanner leverages advanced photon-counting detector technology developed originally through particle physics research at the European Laboratory for Particle Physics (CERN), home of the Higgs boson discovery. MARS Bioimaging holds the exclusive licence from CERN to commercialise this Medipix3 technology for medical imaging of extremities at the point-of-care and for mobile scanners for clinical research.
About MARS Bioimaging
MARS Bioimaging Limited is a Christchurch, New Zealand–based medical imaging company developing mobile imaging technology that delivers premium imaging in community-based settings. MARS imaging systems leverage advanced Medipix detector technology developed at the European Laboratory for Particle Physics (CERN) by the Medipix3 Collaboration, which includes the University of Canterbury. MARS Bioimaging holds an exclusive CERN licence to commercialise Medipix3-based photon-counting CT scanners for extremity imaging in point-of-care settings (and for small-animal imaging) in its defined markets.


 Commerce Commission: Baseline Research Report On The State Of Competition In New Zealand
Commerce Commission: Baseline Research Report On The State Of Competition In New Zealand University of Auckland: Junk Food Designed To Make Us Eat More, Study Finds
University of Auckland: Junk Food Designed To Make Us Eat More, Study Finds Spark: New Report Sets Out Outcomes-Led Approach To Lift Rural Connectivity Using The Right Mix Of Technologies
Spark: New Report Sets Out Outcomes-Led Approach To Lift Rural Connectivity Using The Right Mix Of Technologies Bill Bennett: Fixed Voice Rules Head For Deregulation
Bill Bennett: Fixed Voice Rules Head For Deregulation UN Department of Global Communications: United Nations Proposes New Global Dashboard To Measure Progress Beyond GDP
UN Department of Global Communications: United Nations Proposes New Global Dashboard To Measure Progress Beyond GDP Banking Ombudsman Scheme: Fraud Check Delays Well Worth The Inconvenience, Says Banking Ombudsman
Banking Ombudsman Scheme: Fraud Check Delays Well Worth The Inconvenience, Says Banking Ombudsman


